Blog: Brian Cooke & Bruce Munday. Featured image: Ron Sinclair.
Chasing the DNA trail has become something of a 21st century family sport. So it is hardly surprising that researchers are interested in the genomics of that great little breeder, the wild rabbit. But first – some history.
Introductions
In 1859 Thomas Austin at Barwon Park in western Victoria received from his brother in England 66 partridges, a couple of dozen rabbits and a few hares. The Geelong Advertiser lauded this acquisition but with caution: ‘We trust that Mr Austin will use due caution in keeping his hares in proper duress, otherwise they will become as destructive a pest to the farmer here as they are in the old country’.
Wise words but the wrong target, the hares dying from heat stress within a fortnight, the rabbits thriving such that Austin could soon boast ‘the English wild rabbit I have in thousands’.
Austin’s consignment of rabbits began the journey as 6 European wild rabbits and 7 ‘domestic’ rabbits sourced from local villagers. It numbered 24 on arrival, a foretaste of what was to come.
|
Domestic and wild rabbits are descended from a species of wild rabbit from central and north-eastern Spain Oryctolagus cuniculus cuniculus, which is closely related to Oryctolagus cuniculus algirus now found in Portugal and southern Spain. The domestic strains of rabbits have been selected and ‘inbred’ to emphasise specific characteristics like larger size or coat colour. They generally have less genetic diversity than wild rabbits, less natural fear of predators, and they no longer burrow. |
By 1867, eight years after Austin’s initial release, rabbits in large numbers were reported up to 60 km from Barwon Park. Four years later again they were found at Donald, 270 km north, but this time delivered by a couple of pastoralists who had been gifted rabbits by Austin. Yes, Austin was generous with rabbits to his neighbours who (at first) valued them for sport. Colonists probably seeded rabbits well ahead of their natural spread, although history also records observations of rabbits spreading annually like a wave across the landscape in various parts of Australia.
In 1988, CSIRO scientists Eleanor Stodart and Ian Parer analysed newspaper reports, government documents and accounts from diaries to ascertain when rabbits were first recorded in different localities across Australia. From this they detailed the spread of introduced invasive European rabbits. They confirmed that, although rabbits had been imported many times and established locally in many places, few, if any, were sufficiently robust to spread until the release made by Thomas Austin.
Peacock and Abbott (2013) added to that story with records of over 300 rabbit introductions or relocations in Australia, more than 90 of them prior to the 1859 release at Barwon Park. Most mainland releases failed although there were exceptions, such as Ellenthorpe (Tasmania, 1859), Anlaby Estate (SA, 1857-8), Tempe (NSW, 1860s) and numerous locations in Victoria in the 1860s ‘likely sourced from Barwon Park’. On the other hand, many releases on islands around Australia persisted, probably owing to the absence of native predators such as quolls and goannas that were present on the mainland.

Barwon Park and Anlaby Estate were sites of early rabbit releases. Images: Bruce Munday & State Library of SA.
The type and source of all the introductions of rabbits recorded by Peacock and Abbot is unknown. It is thought that many of the rabbits introduced to Australia were domestic types because they were easier to manage on long voyages. Their domestic features, such as living above ground, would have limited their suitability to life in the wild. It also appears that there were at least some other importations of wild rabbits.
As an example, sometime after 1860, Thomas Holt, a wealthy NSW pastoralist, released and maintained rabbits around his residence ‘The Warren’ overlooking Cook’s River in Sydney. A newspaper article from 1865 noted that: ‘The English breed of wild rabbit has found a suitable home there, and from sixty, the original stock, they have increased to several thousands, although a great many have been shot during the last winters.’ The original source of these rabbits is unknown. Successful introductions where local populations established, such as those at Anlaby Estate, may have also had wild characteristics.
Genetics and other factors
Deciphering historical records of the rabbit’s spread must contend with the uncertainty of whether rabbits spread naturally or were simply taken to new localities by settlers. However, aligning historical records with modern genetic information is helping to paint a better picture of the astounding spread of rabbits across vast areas of temperate and sub-tropical Australia, and their subsequent devastating effect.
Understanding rabbit genetics and their pattern of colonisation is more than an act of curiosity. It may reveal secrets to the varied response and genetic resistance of rabbits to diseases like myxomatosis and calicivirus, enabling better rabbit control. Understanding dispersal rates for rabbits and their genes will also help in assessing the likely spread of gene-drive bio-controls, should they ever become a possibility.
To achieve a better understanding of rabbit genetics and distribution requires knowledge of:
- How rabbits spread
- How gene exchange occurs between distinct rabbit populations
- Evolutionary change
- The role of environmental influences.
How rabbits spread.
Rabbits exist in social hierarchies that are dominated by a male and a few females. They are usually bound to their warren – their primary ‘safe place’ and maternity ward. Few studies have documented their movement but the evidence to hand is that it is usually younger rabbits, especially males, who leave the warren in search of a new social group (Parer, 1982). The rate of movement is likely to be higher when food is scarce.
Historic evidence shows that starving rabbits will move considerable distances in times of drought, and some historic expansions occurred during periods of widespread drought (Deacon 1953). While we know something of how modern territorial rabbits behave when bounded by other rabbits, their nature when in a frontier situation is open to speculation.
How gene exchange occurs between distinct rabbit populations.
Until recently, we had no clear picture of the likely genetic outcome when an expanding or introduced population of rabbits met a well-established population of a different genotype. However, computer modelling shows that genes from the established population would remain common simply because the first arrivals from a spreading population mate with those already present rather than displacing them. Any selective advantage of the invading genotypes can change the pattern, but only after thousands of generations offset the predominance of genes from the initial population.
Studies on gene-flow across the stable hybrid zone between the two sub-species of Oryctolagus also show there is no simple merging of genotypes, strong elements of both persisting as genes are interchanged in either direction.
The extent of genetic introgression from one population to another is influenced by many factors such as the relative size of each population, or the time a resident rabbit population had been established.
Evolutionary change.
Evolution (natural selection) potentially enables domestic rabbits to better adapt to their environment. For example, domestic chinchilla rabbits released on Spilsby Island showed evidence of reverting to ‘agouti’ fur colour forty years later (personal observations). There is also good evidence of intense selection for fur colour in mainland Australia (Stodart, 1965) with ‘yellow’ rabbits that lack melanin being common in hot inland areas and black rabbits being common in high altitude or colder wetter areas.
Rabbits also responded to myxomatosis surprisingly quickly with selection for genetic resistance being evident within only two years of the virus release. Thus, rabbits from a release far distant from Victoria could have been relatively well adapted to their habitat by the time rabbits with genes derived from Barwon Park rabbits arrived.
Environmental influences.
It apparently took 40-50 years for rabbits originating from Barwon Park to reach the New South Wales coast (Stodart and Parer, 1988) as they were not well adapted to the cool, wet forests of the Great Divide. For the same reason, the spread of rabbits west from the Sydney region would have been restricted.
Rabbit populations are more structured genetically in coastal Australia than in the drier inland regions (Fuller et al. 1996) presumably because the climate is milder, and there is no massive mortality during droughts. Territories are smaller and social barriers to the spread of rabbits are more strongly maintained.
To summarise what has been long understood:
- History tells us that there were many attempts to introduce rabbits across Australia. While many failed, some rabbits survived and some populations flourished. Barwon Park was a very ‘successful’ introduction that flourished, largely due to their ‘wildness’, and led to the spread of rabbits by expansion of their range and by transport and ‘seeding’ by colonists.
- Genetics tells us that an exchange of genes is likely when different populations of rabbits come into contact, and that by breeding with their neighbours genetic exchange can spread further without the necessity of rabbits migrating themselves. Over time, evolutionary change can also occur as rabbits are selected for new environments.
Modern genetic studies
Recent genetic research is helping us develop a better understanding of the spread of rabbits in Australia.
In 2019 Iannella et.al. recorded regional differences in the genetics of rabbits across Australia, and concluded it supported the historic evidence of numerous rabbit introductions. In 2022 Alves et.al. showed that genes from the original Barwon Park rabbits were evident across Australia and derived from just five females, but those genetics were not present in all places, e.g. being absent in rabbits from Sydney and Cattai, and rabbits in Tasmania derived primarily from domestic stock. Both studies were limited by access to genetic samples and there are some big geographic gaps in understanding. Further sampling may reveal more local populations like those of Sydney and Cattai.
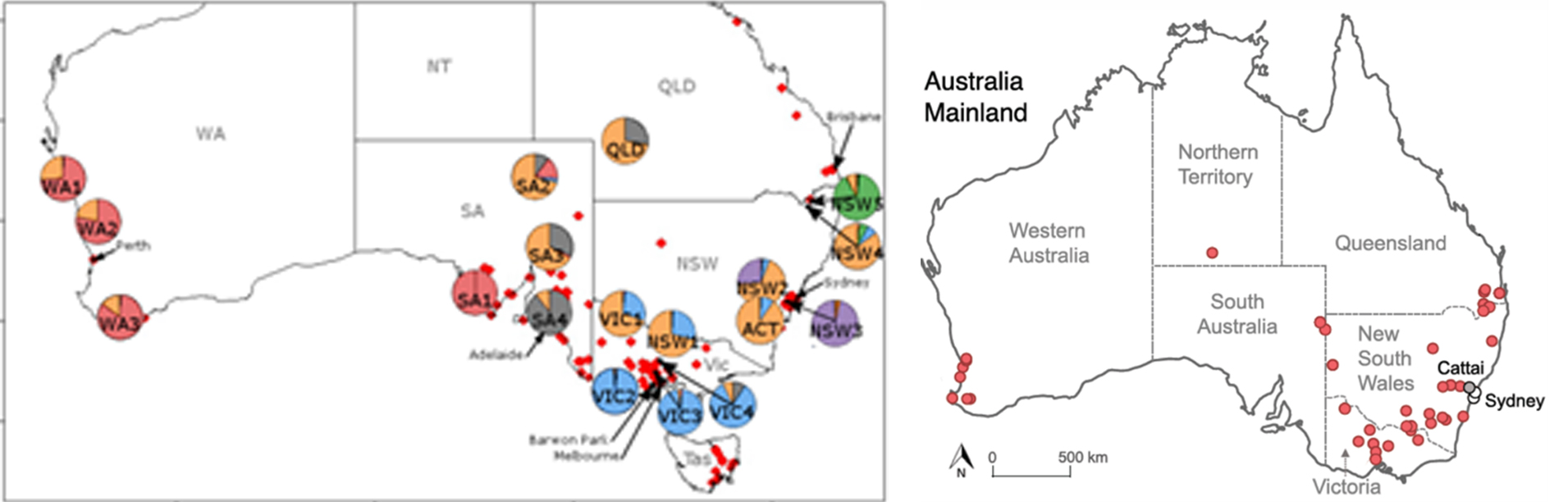
Maps of sample sites from Iannella et.al (2019) and Alves et.al (2022)
This background information makes it more understandable why rabbits around different colonial centres in Australia retain distinct genetic patterns that reflect their original genetic heritage, and elements of the invasive rabbits that spread from Austin’s release of wild stock. It is likely that genes from previously established populations, as well as those from wild rabbits, played a role in helping rabbits adapt and meet the challenges of living in Australia’s interior.
Future research
Combining historic and genetic evidence is producing a clearer picture of rabbit introductions, survival and spread throughout Australia, although still with numerous gaps in geographic coverage and understanding. To fill those gaps and obtain greater clarity will require further investigations, such as:
- Genetic sampling and studies. Further analysis of the type reported by Iannella et al. (2013) and Alves et al. (2022) backed by wider genetic sampling and analysis to fill the gaps in information from all parts of the continent and advance our overall understanding.
- Climate. Climate broadly determines rabbit social structure (territoriality and social group size) and consequently genetic population structure (Fuller et al. 1996). This needs to be considered in a regional sense (i.e. humid coastal regions compared with arid inland areas), and it will also be useful to better understand the relationships between rabbit populations and environmental limits (especially temperature and humidity).
- Historic ecology. Dramatic changes to Australian landscapes are also likely to have played a part in the survival and spread of rabbits, making the country more amenable to their survival and invasion. Reductions in the distribution and abundance of native predators and the impact of pastoral and agricultural development on ‘improving’ the extent and quality of feed available to rabbits could both have been significant.
- Case histories and conceptual models. Other useful enquiries include case histories as a way of teasing out details and building conceptual models that take account of rabbit ecology as well as historical records and genetic data.
- Comparative studies. Comparative studies involving other vertebrate species may also be helpful for interpreting genetic data and the introgression of genes from existing populations into colonizing ones.
Finally, it is useful to remember that here we have a good example of how progress in science is made. In posing an alternative to the ideas of Stodart and Parer (1988), the paper by Iannella et al. (2019) helped frame and sharpen the questions asked in the subsequent paper by Alves et al. (2022) and build a clearer picture of how invasive rabbits colonized Australia, while posing yet more questions to be addressed by further research and investigation.
We can conclude from these recent studies that the significance of Austin’s rabbits or other rabbits released by European settlers partly depends on scale, both temporal and spatial. At a local level around Australia’s early European population centres, wild rabbits might have had little impact on colonies that were already established (e.g. at Cattai), however, taken at a continental level, genes from Austin’s rabbits were important in catalysing the wave of rabbits that eventually colonized two thirds of Australia.
Bruce Munday’s book, Those Wild Rabbits – How they shaped Australia, highlights not only the damage done and the mostly futile efforts at rabbit control but also Australia’s missed opportunities. “After years of shooting, trapping, poisoning, and habitat destruction, the real breakthroughs eventually came from research” says Munday. “Research identifying the rabbit’s rare points of vulnerability”.
“Informed and tested knowledge are essential for an effective defence against this determined pest.”
References
Anon. (1865) Mr. Thomas Holt, The Illustrated Sydney News , 16 May 1865, page 4.
Currat, M., Ruedi, M., Petit, R.J. and Excoffier, L. (2008). ‘The hidden side of invasions: Massive introgression by local genes.’ Evolution, 62: 1908-1920. https://doi.org/10.1111/j.1558-5646.2008.00413.x
Deacon, E.L. (1953). ‘ Climate change in Australia since 1880’. Australian Journal of Physics, 6, 209-218.
Fuller SJ, Mather PB, Wilson JC (1996) ‘Limited genetic differentiation among wild Oryctolagus cuniculus L. (rabbit) populations in arid eastern Australia’. Heredity 77, 138–145.
Iannella, A., Peacock, D., Cassey, P. et al. (2019). ‘Genetic perspectives on the historical introduction of the European rabbit (Oryctolagus cuniculus) to Australia.’ Biological Invasions 21, 603–614. https://doi.org/10.1007/s10530-018-1849-2
Munday, B. (2017). ‘Those Wild Rabbits – How they shaped Australia’. Wakefield Press, Adelaide.
Parer, I. (1982) ‘Dispersal of the wild rabbit, Oryctolagus cuniculus, at Urana in New South Wales.’ Australian Wildlife Research 9(3): 427-441.
Peacock, D. and Abbott, I. (2013) ‘The role of quoll (Dasyurus) predation in the outcome of pre-1900 introductions of rabbits (Oryctolagus cuniculus) to the mainland and islands of Australia’. Australian Journal of Zoology, 61, 206-280.
Quinn, C.B., Preckler-Quisquater, S., Akins, J.R. et al. (2022). ‘Contrasting genetic trajectories of endangered and expanding red fox populations in the western U.S.’ Heredity 129, 123–136. https://doi.org/10.1038/s41437-022-00522-4
Stodart, E. (1965). ‘A study of the biology of the wild rabbit in climatically different regions in eastern Australia. III. Some data on the evolution of coat colour.’ CSIRO Wildlife Research 10, 73 – 82.
Stodart, E. and Myers, K. (1964). ‘A comparison of behaviour, reproduction, and mortality of wild and domestic rabbits in confined population’. CSIRO Wildlife Research 9, 144 – 159.
Stodart, E. and Parer, I. (1988). ‘Colonisation of Australia by the rabbit Oryctolagus cuniculus (L.).’ Canberra: CSIRO; https://doi.org/10.4225/08/5a5cec3187bd7

